Research Project 8
State of the art
- Due to the ability of the Lewis basic sulfur atom to poison catalysts C-S bond functionalization has remained a challenge.
- Most reactions involve C-C bond formations; identifying new reactions such as mild reduction would be fruitful in chemo- and regio-selective manner.
- Identifying new organic scaffolds as non-toxic, inexpensive and eco- friendly photocatalysts has recently become a dynamic research area. The development of accessible and cheap metal-free photocatalysts is still highly desirable.
- Creating chiral centers have been achieved by photoredox chemistry but only few enantioselective processes are documented. Competent chiral organic photocatalysts are even less described.
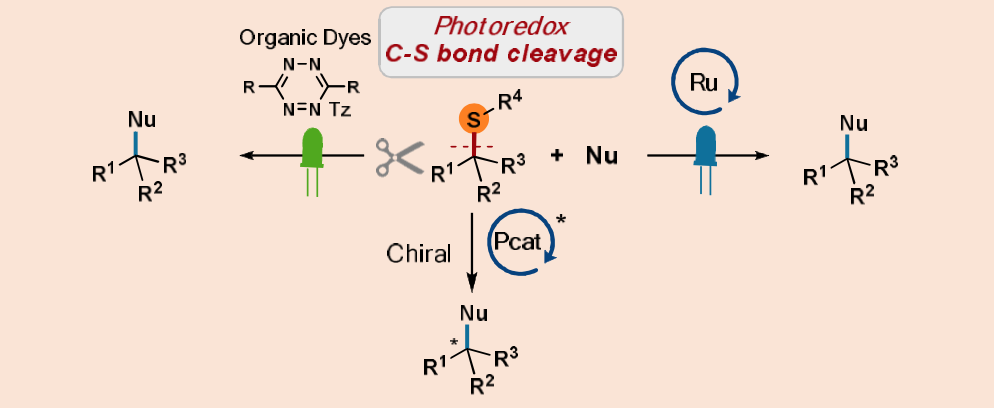 Scheme 8. Photocatalytic C – S bond cleavage.
Scheme 8. Photocatalytic C – S bond cleavage.
Progress beyond the state of the art
- A cleavage of sulfidic C-S bonds under visible-light irradiation was harnessed under neutral conditions to synthesize valuable organic compounds. This approach can be broadened to complex targets (Scheme 8).
- New organic dyes, tetrazines displaying high oxidative ability have been designed and prepared. These compounds are among the smallest photoredox catalysts reported in the literature and can be generated in situ from commercially available compounds. Moreover, their redox properties can be tuned by modifying the substitution patterns to develop new transformations.
Key Objectives & Expected Results
Performance Indicators
- Photoredox-catalyzed C-S bond cleavage will be used as a key synthetic step to synthesize \natural product such as Muchimangins.
- At least one new photocatalytic reductive transformation will be developed.
- At least one chiral organic dye will be designed to carry out
enantioselective photocatalytic transformations.





